Light is becoming a tool against cancer. Doctors now use targeted beams and special light sensitive drugs to destroy tumors while sparing more healthy tissue. From photodynamic therapy to newer photoimmunotherapy and laser based heat procedures, these options aim for precision with shorter recoveries. If you or someone you love is hearing about light based care, this guide explains how it works, who it may help, and what to ask.
In This Article
- How different light based treatments work in plain language
- What a typical appointment and recovery look like
- Benefits and tradeoffs compared with surgery and chemo
- Questions to ask your care team before choosing
- How to prepare and care for yourself after treatment
Light Based Cancer Treatments Explained
by Alex Jordan, InnerSelf.comYou hear the word light and it sounds gentle. Cancer treatment does not. Putting those words together can feel confusing at first. Yet in clinic rooms around the world, doctors are using carefully chosen wavelengths to activate drugs, heat tiny areas of tissue, and even flag tumor cells for the immune system. This is not sci fi. It is a practical shift toward precision, where the goal is simple. Hurt the tumor. Spare the person. Let’s walk through what that really means for you.
How Light Unlocks Precision
Think of light as a key that only fits certain locks. In photodynamic therapy, often called PDT, you receive a drug that likes to sit in tumor cells more than in healthy cells. On its own, the drug waits quietly. When a doctor shines a specific color of light on the area, the drug wakes up and makes a burst of reactive oxygen inside those cancer cells. That burst damages the cells from within, like popping tiny balloons where you want them to pop. Because the light only reaches a defined spot, the effect is local and timed.
Photoimmunotherapy takes the same idea one step further. Instead of a drug that passively sits in a tumor, the medicine is attached to a targeting antibody that recognizes a marker on the outside of cancer cells. After the drug finds its target, the doctor uses near infrared light to activate it. The hit to the tumor cell is direct, and the mess left behind can call in the immune system to learn what to attack next. You can think of it as turning the tumor into a teaching moment for your own defenses, with light acting as the on switch.
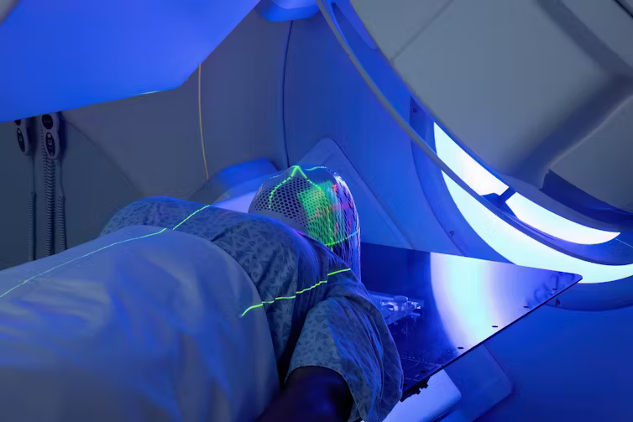
Laser based heat treatments work differently. In laser interstitial thermal therapy, or LITT, a surgeon threads a fine laser fiber into the tumor through a small opening. Imaging guides the tip to exactly the right spot. Then the laser produces focused heat that cooks the tumor while software monitors the temperature so nearby brain or other delicate tissue stays within safe limits. When heat is the tool, there is no special drug to activate, just physics and careful control.
There is also photothermal therapy at the research and early clinical edge. Here, light absorbing particles or dyes gather in the tumor, and a beam of light warms only the areas that hold those particles. The warmth is enough to damage cancer cells but not so high that it harms the tissue around them. The common thread across all of these is selectivity. Light gives clinicians a way to draw a boundary, then press go.
What Patients Actually Experience
The visit depends on the method, but the flow shares a pattern. For PDT and photoimmunotherapy, your team starts with a review of your medications, allergies, and the tumor’s location. You receive the light sensitive drug through an IV or sometimes as a topical application if the cancer is on the skin or just under the surface. There is a waiting window while the drug spreads and settles, which can be hours to days depending on the medicine. During this period, you follow light safety instructions to protect your skin and eyes because the whole point is that the drug reacts to light.
When it is time, the doctor shines the light directly on the tumor. In the mouth or throat, a thin fiber can reach tight spaces. In the lungs, a bronchoscope helps guide the light. On the skin, a lamp or small handheld device does the job. You may feel warmth or a prickly sensation as the tissue reacts. Treatments are usually outpatient. You go home the same day with instructions to limit bright light for a short period and to keep the area clean and dry.
Photoimmunotherapy sessions feel similar from your side. The difference is the drug knows its target more specifically, so the activation step is focused on tissue where those targets live. Some people notice swelling where the tumor is, a sign that the treatment caused local injury and fluid shift. Your team will talk about steroids, pain control, and when to call if swelling feels tight or breathing is harder. Most swelling peaks and then fades within days.
LITT is a hospital procedure because it involves imaging and anesthesia. You meet both the surgeon and the anesthesia team. Through a tiny incision and a small burr hole for brain cases, the laser fiber slides into position. Real time MRI or other imaging confirms placement. The heating step is measured in minutes while the team watches thermal maps that show color changes as tissue warms. Recovery is faster than open surgery for many patients, but you still rest, manage soreness, and watch for headaches, nausea, or neurological changes. Many people go home within a day or two.
Where The Science Stands Now
Light based care is not a single treatment but a toolbox. Some tools are already standard for certain cancers. PDT has long been used for early skin cancers, pre cancers in the esophagus, and certain lung lesions because light can reach them directly. As instruments get better, PDT is showing up in more places where doctors can safely deliver light. Photoimmunotherapy is newer. It builds on years of antibody therapy by adding a light trigger. Early use focuses on tumors that display known surface markers, such as some head and neck cancers. Because the approach is targeted and local, researchers are testing it in combination with systemic treatments like checkpoint inhibitors to see whether local tumor destruction can train wider immune responses.
Heat based laser therapy has special value where open surgery is risky or recovery would take too long. In the brain, for example, a small pathway for a laser fiber avoids a large opening, and real time maps help avoid critical regions. That does not make it simple. Tumors can be diffuse, and heat falls off with distance, so success depends on precise planning. In the spine and liver, similar principles apply. The promise is control with smaller scars and shorter hospital stays for carefully chosen cases.
Across the board, what is changing fast are the ways doctors guide and monitor the light. More flexible fibers can snake into smaller spaces. Imaging now overlays temperature maps on anatomy so surgeons see where heat is and is not. New light sensitive drugs are being designed to find different tumor markers and to use colors of light that travel deeper through tissue. Together, these advances move light based care from niche to option, especially when the goal is to shrink a tumor, open a blocked airway, reduce pain, or buy time for other therapies to work.
It is also worth saying plainly what these treatments are not. They are not magic bullets. They work best when the tumor is in reach of light or a laser probe, when the biology matches the target, and when your overall health supports recovery. They can play the lead role or support other treatments by debulking a mass, unblocking a passage, or teaching the immune system what to look for.
Limits Risks And Tradeoffs
Every treatment asks you to weigh benefits against risks. With light based options, the benefits include precision, less damage to healthy tissue, repeatability, and shorter recovery. Because the light does not linger like a drug in the bloodstream, side effects are often local and time limited. Many people return to daily routines sooner than they expected.
The limits reflect the same physics that make light attractive. Light does not travel far through dense tissue. If a tumor sits deep without a safe pathway, your team may not be able to get enough light to it without harming the skin or the structures in between. That is one reason laser fibers and scopes are so important. They carry the light to the problem.
Risks vary by method. PDT and photoimmunotherapy can cause temporary sensitivity to sunlight and bright indoor light. You will get specific instructions about clothing, sunglasses, and how long to be cautious. The treated area can be sore, swollen, or ulcerated briefly as damaged tumor tissue breaks down. In narrow spaces like the throat, swelling can be uncomfortable, and your team will plan ahead to control it.
Laser heat carries risks typical of small surgical procedures. Infection, bleeding, or damage to nearby structures are possible but uncommon when teams follow careful protocols. In brain procedures, swelling can temporarily worsen symptoms before they improve. That is why close monitoring after treatment matters. For spinal or liver targets, pain control and watching for fever or weakness are part of aftercare.
Another tradeoff is expectations. Light based care is very good at local control and symptom relief. It is not always a cure, especially for widely spread disease. Framing the goal clearly helps you judge success. If the aim is to reopen an airway so you can breathe, or reduce pain by shrinking a mass that presses on nerves, you can feel the difference quickly and meaningfully even while other treatments continue in the background.
How To Talk With Your Care Team
Good decisions grow from good questions. Here are ones that help you and your clinicians decide whether light based therapy fits your situation. Where is the tumor and how will you get the light there. What drug or device will you use and why. How many sessions should I expect. What is the goal in my case relief, control, or a path to another treatment. What side effects are most likely for me given where the tumor sits. What signs at home mean I should call or go in.
Ask about preparation. For light sensitive drugs, you will plan clothing, sunscreen, and timing for errands to avoid accidental activation. For laser procedures, you will plan rides, a short hospital stay, and time off work. Clarify every medication you take, including supplements, because some interact with anesthesia or affect bleeding risk. If you use a CPAP, bring it. If you manage diabetes, ask how to handle food and insulin around the procedure time.
Talk about recovery in specifics. When can you shower. When can you eat normally. What level of soreness is expected and what is not. If the treatment is in your mouth or throat, how should you handle eating and drinking while things heal. If the treatment is in your brain or spine, what activities should you avoid and for how long. Precise answers reduce fear because you know what you are aiming for week by week.
Finally, ask how light based therapy fits into the larger plan. Will it be combined with immunotherapy, chemotherapy, radiation, or surgery. If so, in what order and with what goals. The more you understand the overall arc, the easier it is to ride the ups and downs of each step without losing sight of why you are doing it.
About the Author
Alex Jordan is a staff writer for InnerSelf.com
Recommended Books
The Emperor of All Maladies: A Biography of Cancer
A sweeping narrative of cancer’s history and the hard won advances that shape today’s treatment choices, written in a way that helps patients and families feel informed and empowered.
Article Recap
Light based cancer care uses focused energy to activate drugs, heat small targets, and rally the immune system with precision. Options like photodynamic therapy, photoimmunotherapy, and laser thermal ablation can shrink tumors, ease symptoms, and speed recovery when the tumor is reachable and the goal is clear. Ask practical questions, plan aftercare, and fit light based tools into your overall treatment plan.
#LightTherapy #CancerCare #Photoimmunotherapy